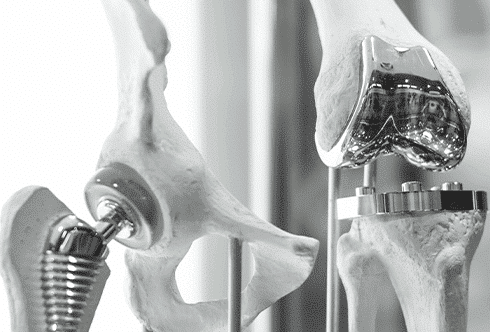
The MedTech sector continues to influence the evolution of orthopedic care through advances in digital technologies, biomaterials, additive manufacturing, and precision-driven surgical workflows. Innovations in orthopedics are increasingly centered on patient-specific treatment strategies that aim to improve implant integration, optimize procedural accuracy, and support long-term functional outcomes. The convergence of engineering, software development, and clinical practice has accelerated the adoption of technologies such as 3D printing, robotic-assisted orthopedic surgery, and advanced imaging-based planning systems. As healthcare systems place greater emphasis on procedural efficiency, regulatory compliance, and outcome-driven care models, orthopedic MedTech remains one of the most active domains of medical innovation. Read more...
Orthopedic Medical Device Translation
from English into French
Orthopedic medical devices require precise, consistent, and regulation-aware communication across every stage of the product lifecycle. From implant documentation and surgical technique guides to IFUs and medical software interfaces, accurate English-to-French translation is essential to ensure patient safety, regulatory compliance, and market access. Specialized orthopedic medical device translation and localization help manufacturers deliver reliable documentation and clear user information for French-speaking healthcare professionals and patients.
Specialized Orthopedic Medical Device Translation
Orthopedic medical device translation requires more than general medical knowledge. Orthopedic implants, surgical instruments, fixation systems, navigation platforms, and rehabilitation technologies all involve highly specialized terminology that must remain accurate and consistent throughout the documentation.*
As a specialized English-to-French medical translator, I provide orthopedic medical device translation services tailored to the MedTech industry. The objective is not only linguistic accuracy, but also clarity, usability, and regulatory consistency across all translated materials.
Orthopedic manufacturers often work with complex documentation intended for surgeons, distributors, healthcare professionals, regulatory authorities, and patients. Precise translation helps reduce ambiguity, support product adoption, and maintain terminological consistency across multilingual content.
French Translation of Orthopedic Device Documentation
Orthopedic medical device documentation includes a wide range of technical, regulatory, clinical, and user-facing materials. Each document type requires a different level of terminology management, readability, and localization. I translate orthopedic medical device documentation from English into French for manufacturers, suppliers, and MedTech companies working with international markets and French-speaking regions.
Typical content includes:
- Instructions for Use (IFUs)
- Medical device labeling
- Surgical technique guides
- Technical documentation
- User manuals
- Training materials
- Clinical and product documentation
- Software interfaces and connected device content
- Marketing and website content for orthopedic products
Specialties: Hip Orthopedics, Knee Orthopedics, Spine Orthopedics, Shoulder Orthopedics, Foot and Ankle Orthopedics, Hand and Wrist Orthopedics, Elbow Orthopedics, Sports Medicine, Trauma Orthopedics, Orthopedic Surgery, Joint Replacement, Orthopedic Implants, Arthroscopy, Pediatric Orthopedics, Musculoskeletal Disorders, Orthobiologics, Rehabilitation and Mobility Devices, Prosthetics and Orthotics
Translating IFUs, Labeling, and Surgical Technique Guides from English into French
Instructions for Use (IFUs) and labeling are among the most sensitive documents in the medical device industry. In orthopedic MedTech, even minor inconsistencies can affect comprehension, usability, and compliance.
Surgical technique guides also require careful terminology management and clear phrasing adapted to French-speaking healthcare professionals. The translation must remain technically accurate while preserving the structure and intent of the original content.
Consistent French terminology across IFUs, packaging, labeling, and technical files contributes to safer communication and a more reliable user experience.
Regulatory Translation for Orthopedic Medical Devices
Orthopedic medical device translation must support regulatory compliance as well as technical accuracy. Documentation intended for the European market often requires alignment with EU MDR terminology, multilingual labeling requirements, and standardized medical language.
Regulatory translation involves particular attention to consistency, traceability, and clarity across all documentation components. This is especially important for orthopedic implants, surgical systems, and Class II or Class III medical devices.
French Translation for EU MDR Compliance
EU MDR-compliant translation requires terminology consistency across IFUs, labeling, safety information, and technical documentation. French translations must remain aligned with the original English source while meeting the linguistic expectations of the target market.
Clear and accurate regulatory translation supports:
- patient safety,
- readability,
- risk communication,
- and smoother international product distribution.
A specialized orthopedic medical translator understands the importance of maintaining precise terminology throughout the entire documentation ecosystem.
Orthopedic Medical Device Localization into French
Medical device localization goes beyond translation alone. Orthopedic MedTech manufacturers and companies increasingly rely on digital platforms, connected technologies, software interfaces, and multilingual communication tools that must be adapted for French-speaking users.
French localization helps ensure that terminology, formatting, navigation, and user-facing content remain natural, clear, and consistent within the target environment.
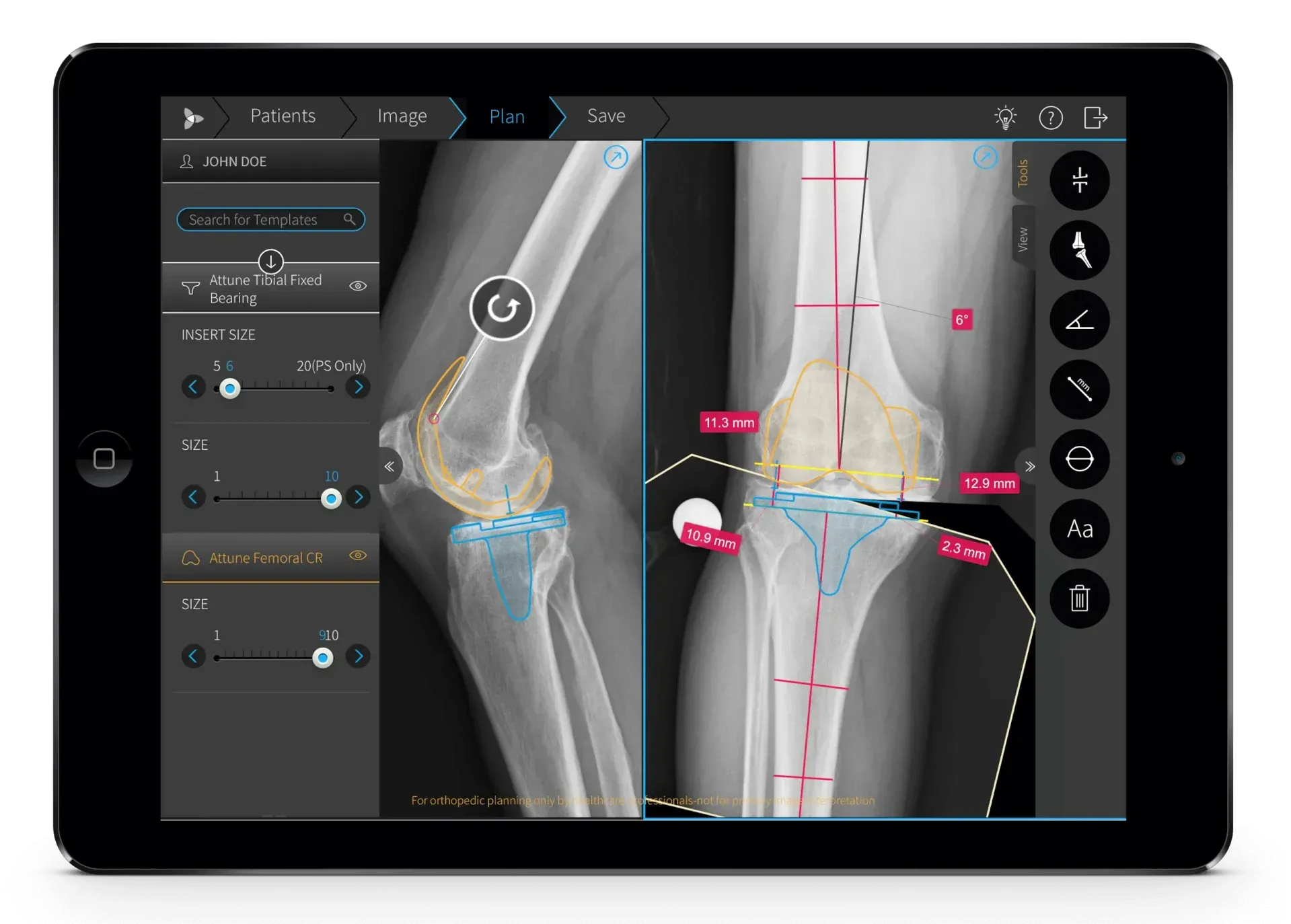
Localization of Orthopedic Surgical Software and Digital Content
Orthopedic software localization may include:
- user interfaces,
- orthopedic surgical planning softwares
- connected device applications,
- e-learning platforms,
- patient communication tools,
- and web-based MedTech content.
Accurate and user-friendly localization improves usability and supports a better experience for healthcare professionals and end users. It also helps maintain consistency between software, documentation, and product labeling. In MedTech, linguistic precision and usability are closely connected.
Why Specialized Orthopedic Translation Expertise Matters
Orthopedic medical translation combines technical language, regulatory requirements, and industry-specific terminology. Working with a specialized English-to-French medical translator with extensive experience in orthopedics helps ensure consistency across all documentation and communication materials.
A focused specialization in orthopedic medical devices also facilitates more accurate terminology research, improved coherence across projects, and a better understanding of MedTech documentation standards.
Terminology Management and Quality Assurance
Terminology consistency is essential in orthopedic medical device translation. Implant names, procedural terminology, device components, and regulatory language must remain stable across all translated materials.
Quality assurance includes:
- terminology verification,
- consistency checks,
- proofreading,
- and attention to formatting and regulatory details.
For orthopedic manufacturers, accurate French translation contributes to clearer communication, stronger documentation quality, and more reliable localization workflows.
Benefits for Orthopedic Medical Device Manufacturers
Specialized orthopedic medical device translation helps manufacturers support regulatory compliance, improve communication with French-speaking healthcare professionals, and maintain consistency across technical and marketing documentation.
Accurate English-to-French localization also contributes to smoother market access, clearer product information, stronger product adoption, and increased sales across French-speaking markets.


I am Emi Lecret, a native French speaker, medical writer, translator, localization specialist, and UX designer with extensive experience supporting pioneering orthopedic MedTech companies and medical device manufacturers. I am passionate about innovative medical technologies and I help translate and localize orthopedic medical device content from English into French with a strong focus on accuracy, regulatory consistency, usability, and clear communication for French-speaking markets.
THEY TRUST ME



For more insights, read this related blog article